|
Illustration by Elisa Visher Carly Rozins presented her work on integrating disease models with a 'perception kernel' that allows vectors to move based upon how they are perceiving resources in their surroundings. For a Wordsworth based, poetry style summary: Pollinators wander lonely as a cloud And float among the flowers in the Italian nation They search for nectar among the crowd, Of their host, a mountain carnation Whether or not they choose to leave Depends on what their biology says they can perceive Continuous as the stars that shine And twinkle in the Milky Way The carnations upon which the pollinators dine Are spread out into an array The pollinators determine their density with a glance Then to the best patch, they dance As the pollinators move, they Transmit anther smut from flower to flower And thus to disease dynamics, perception kernels may Lend some extra explanatory power Carly has mathed-and mathed-with much thought And from her models, insight can be brought So oft when vectors can perceive, And make decisions About when to leave Based upon how to find the best provisions Smut epidemics will spread faster Amongst all the flowers in the pasture Summary by Elisa Visher
22 Comments
Illustration by Kacie Ring Last week, we had Kacie Ring visit us from the Swei Lab at SFSU to present her research on lyme disease in the Bay Area! This week, we've written our summary as a poem! In California, we have a lizard That cannot survive in those northeastern blizzards. The black-legged tick likes to feed on its blood Which carries a toxin that nips Lyme Disease in the bud Lyme burden is lower where these lizards slither Eco-prophylaxis: Nature's a wizard! Poem by Cara Brook
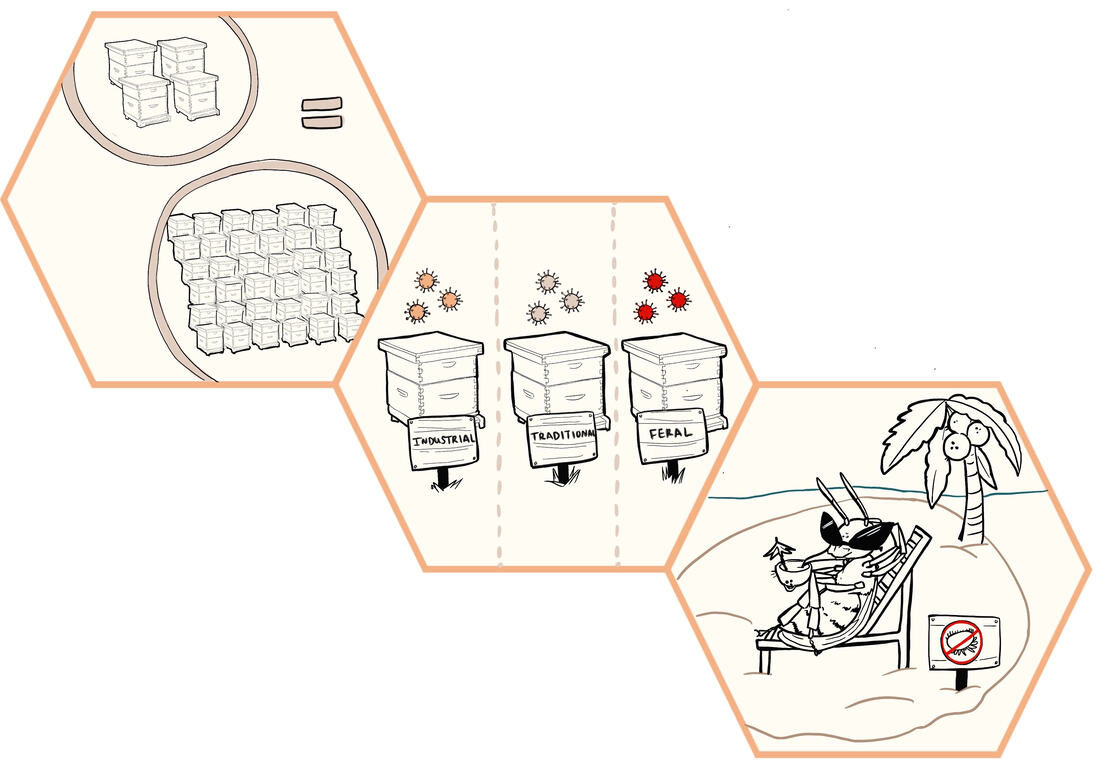
Lewis Bartlett (UGA) presents "Evolutionary Beekeeping – Trade-Offs, Outcomes, and Dangers"12/4/2019 Illustration by Nina Sokolov Lewis has worked at University of Exeter, UC Berkeley, Emory University, and most recently UGA, to understand when and how we can gain novel insights into bee management by incorporating the ecology and evolution of disease. As will come as no surprise to most readers, bee populations (both managed and wild) are in rapid decline. This is due in part to pesticide use, changing land use patterns, and pest/pathogen expansion. In the case of monocropping, there is often a large burst of flowering (i.e. resources) at one point in time, followed by an absence of flowers for the rest of the season. For honey bee managers, this means they need many hives to take full advantage of the bloom, but are then stuck with hungry bees once the flowering is finished. This has, of course, led to movement of bees around the landscape. Such movement has had predictable impacts on the spread of pests and pathogens, both for the managed bees and for the wild populations with which they share habitat. Approximately 3/4 of the Honeybees in North America are moved around the country on trucks. Such industrial-scale bee keeping has brought with it numerous new challenges, especially in terms of disease spread. However, the silver lining of this is that new findings and emerging best practice in bee keeping can be rapidly implemented. In a recent ecological model (see here: https://besjournals.onlinelibrary.wiley.com/doi/full/10.1111/1365-2664.13461), Lewis and colleagues explored how hive orientation might impact pathogen spread by examining the impact of spatial structure on R0 (the number of secondary infections that result, on average, from a given infection in a susceptible population). As expected, prevalence (proportion infected) increases with increasing R0. They then find that the least intensified apiary (a linear array of about nine hives) has a lower predicted prevalence across the spectrum of R0 values than the intensified apiary (with 100s of hives in a grid), but the impact is not particularly strong. This is especially true at higher R0 values (where most estimates for bee pathogens exist). So, it seems like crowding alone can not explain the increased spread of disease being observed. Now broadening the focus beyond whether a bee is infected or not, Lewis turned his attention towards severity of infection. The problem, however, is that this will be affected by numerous inter-related factors, including pesticides, stress, and ecology. As a first test of this, Lewis brought feral (no management), traditionally managed, and industrially managed bees into a common garden for a year, then screened for viruses. He found some grouping of viruses by beekeeping history and higher viral titres on average in highly managed bees. However, feral bees carried much higher titres of many particular viruses. Finally, in terms of the much-hated varroa mites, which have recently emerged and wreaked havoc across the globe, previous work has shown that the presence/absence of the mite changes the prevalence of viral pathogens. As varroa spread across New Zealand, deformed wing virus increased in prevalence and severity (number of copies observed per bee). A similar pattern was observed in Hawaii. Interestingly, strain diversity rapidly decreased as well, leading to a single highly virulent strain dominating the landscape. This is most likely due to the success of this virus in the mite itself (see Campbell et al. 2016 for an elegant demonstration of this). Overall, it seems intuition does not get us far in this system - in part because of the many factors interacting to shape the ecology and evolution of disease in managed settings. Thank goodness for guidance from theory, diligent researchers, and people like Lewis who choose to spend their intellect and time focusing on solving these critical questions! Summary by Britt Koskella
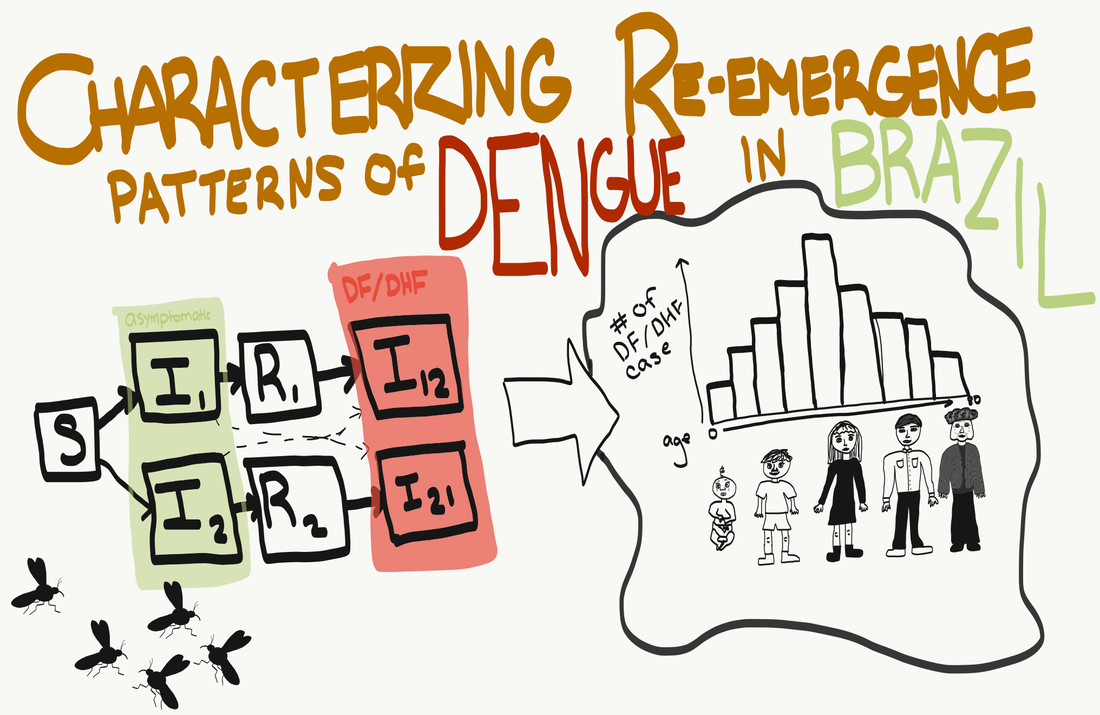
Doodle by Elisa Visher This October, post-doc Saki Takahashi visited our seminar from across the bay to present her work on "Characterizing re-emergence patterns of dengue in Brazil". Saki recently started her post-doc after finishing up her PhD with Jess Metcalf at Princeton. Saki is broadly interested in how immunity drives transmission dynamics in multi-strain pathogen systems. For many infectious diseases, we know that hosts develop immunity after infection. However, this immunity can vary in how long-lived it is, how strong it is, and how broad it is, with all of these variables shaping how immunity influences disease dynamics. Saki uses modelling techniques to elucidate patterns of transmission from epidemiological data. Specifically, Saki build these models with age-structured case incidence data, as it can be especially informative in teasing apart patterns of immunity, strain dynamics, and transmission. This age-structured data can be important because shifts in the age distribution of cases can indicate changes in the distribution of susceptibles and the force of infection of different strains. For the project that she presented to us at the Berkeley EEID seminar, Saki looked at dengue dynamics in Brazil. Dengue is an arboviral disease that has recently re-emerged in Brazil. Globally, it is the most rapidly spreading arboviral disease because of its multiple serotypes, increased dispersion of its vector, urbanization, and global travel. Dengue has four antigenically distinct but interacting serotypes. Humans can be infected by multiple of these serotypes because infection with each strain does not offer complete immunity against others. However, previous infection with a different stain of dengue does change the clinical symptoms of infection. People normally have worse symptoms during their second infection (including dengue fever and dengue hemorrhagic fever), after generally asymptomatic primary infections. In Thailand, where all four strains are endemic, dengue is generally considered a childhood disease because each strain’s force of infection is high enough that people tend to be infected by multiple strains and have clinically symptomatic dengue infection during childhood. However, Thailand has recently had a demographic transition with people living longer and having fewer children, which has decreased this force of infection and led to the average age of cases being older. This illustrates how the age-structured case data that Saki uses can be hugely informative for understanding the transmission dynamics of multi-strain pathogens like dengue. Saki, therefore, turned her attention to using such age-structured data to model dengue dynamics in Brazil, where dengue has been re-emerging since 1986 due to the re-emergence of its mosquito host, Aedes aegypti, in the 1970s. Recently, in 2007, clinical cases of dengue have shifted from mostly being seen in adults to mostly being seen in children. Saki therefore wanted to ask whether she could build a model to capture dengue’s reemergence and temporal variation of transmission in Brazil and use it to identify factors that could explain the shift in the age-distribution of cases. Saki and colleagues built models fit to age-structured epidemiological data which used time varying forces of infection to allow for the unequal circulation of serotypes over time, accounted for the different clinical symptoms of primary and secondary infections, and allowed for differences in reporting rates. The models could also capture finer spatial scales, such as regions within states. From these models, they were able to test between hypotheses to clarify possible causes of the demographic shift in dengue epidemiology over time. They find good support that the shift in the age distribution of cases was driven by the re-emergence of the dengue 2 serotype in 2007, especially in Brazil’s north east states. Basically, before the re-emergence of dengue 2, children likely had asymptomatic first infections, but did not have secondary infections leading to clinical manifestations until adulthood. After 2007, the renewed circulation of dengue 2 led to increased forces of infection and individuals were getting infected with their second serotype of dengue during childhood. In summary, data on the age distribution of cases can be used to yield insights into population susceptibility and disease dynamics. Models using this data suggest that the 2007 shift in the age distribution of cases in Brazil was due to the re-emergence of dengue 2. This may suggest that age-dependent clinical guidelines should developed to reflect changing age burdens. Finally, Saki thinks that one of the next steps for this project is to validate the model with environmental variation data, more epidemiological surveillance data, and more orthogonal data. She also wants to do simulations and modelling on time to endemicity to get an understanding of the epidemiological transmission from reemergence to endemicity. Summary by Elisa Visher
|
AuthorBerkeley EEID Group Archives
February 2020
Categories
All
|
 RSS Feed
RSS Feed
