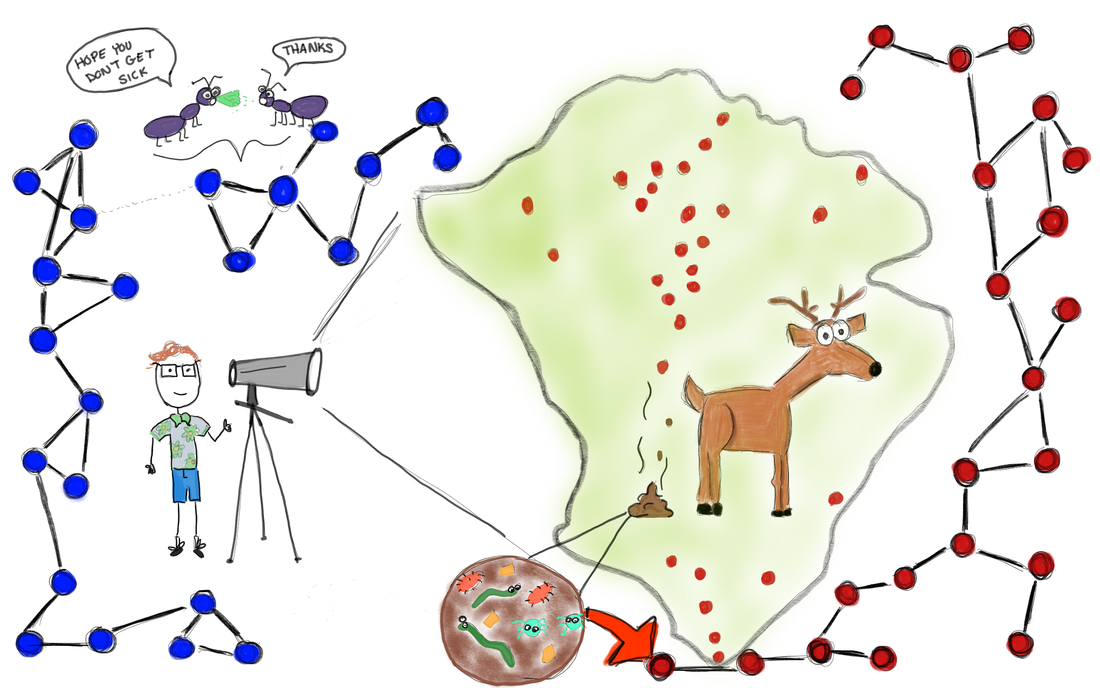
Greg Albery (Georgetown) presents “Social networks, spatial networks, parasites, and deer"10/29/2019 Illustration by Carly Rozins A few weeks ago in the Berkeley EEID seminar we had Dr. Greg Albery present some of his ongoing research. Greg is a recent addition to the Bansal Lab at Georgetown University, but this month he is visiting the Boots lab here at UC Berkeley. Greg’s seminar was on ‘Spocial Networks’. What is a spocial network you might ask? Well this is an important question and a topic often overlooked. But first we need to discuss social networks. A network is a collection of nodes (dots) and edges (lines connecting nodes). Nodes represent hosts and the edges represent interactions, ones that might lead to infectious disease transmission. Networks capture host heterogeneity and provide a rich dataset for determining the effects of sociality on disease dynamics and life history characteristics. Importantly, networks can be empirically derived from surveys, observations, pit tags and radio proximity collars that capture/log social contacts between individuals in the population. These empirical networks are classified as social networks. Therein lies the problem. Since space affects everything, how can these networks be classified as solely social? If space is ignored, it is impossible to determine whether the observed social effects on disease dynamics are actually due to social events, or whether they are truly due to environmental (spatial) constraints. The environment will shape how populations structure and population structure affects social behavior. Greg unraveled these two confounding factors. He collected both spatial and social data for a population of red deer on the Isle of Rum. With an Integrated Nested Laplace Approximation Linear Mixed Models (wha… that was a mouthful!) he determined that spatial heterogeneity is an important factor in both immunity as well as parasitism. Interestingly, he found fine scale spatial patterns in parasitism. Essentially, the best models are spocial, those that control for both spatial and social factors. However, Greg is not done yet! He is now working with an incredibly detailed ant data set on social and spatial behavior and he is on the lookout for even more data. So, if you know of any datasets that include any combination of social, spatial and disease data, Greg would like to chat with you! Summary by Carly Rozins
15 Comments
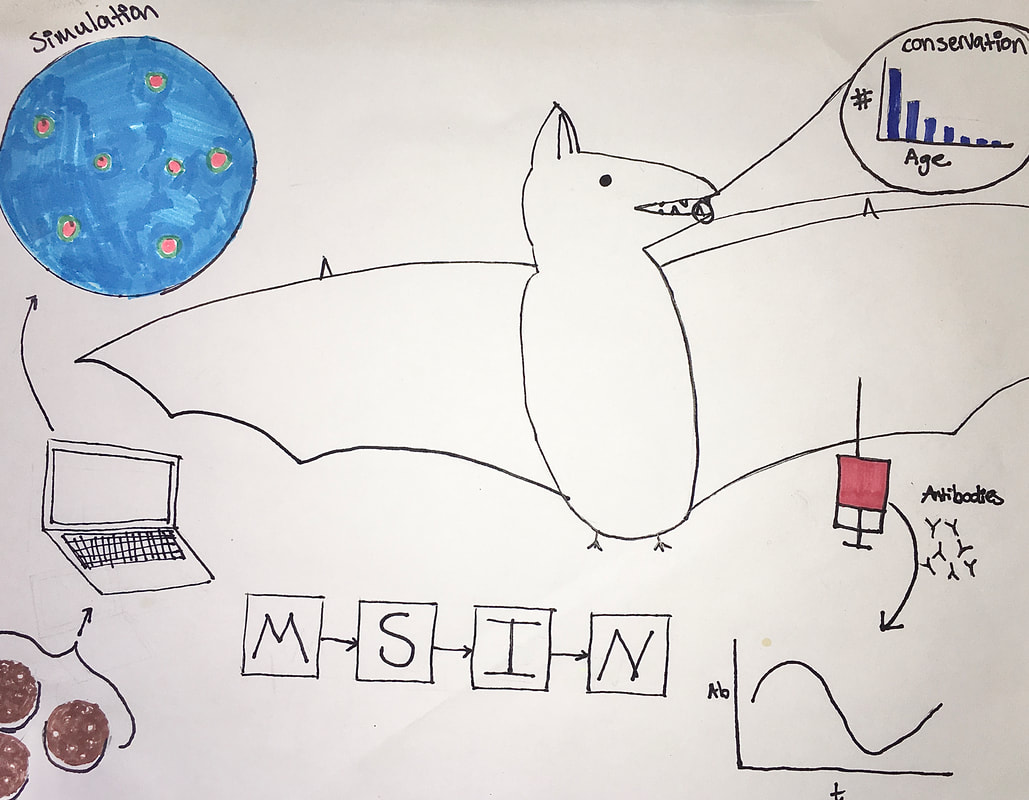
Illustration by Laura Alexander Bats play host to some of the gnarliest, deadliest viruses known today, including those that cause Ebola, Marburg, SARS and MERS. So what gives? Are these fuzzy flying mammals special in some way in their propensity to produce zoonotic pathogens that infect us humans? There’s a growing consensus that the answer to this question is almost certainly “yes!”, but what is it that makes bats special viral vessels? This week, Cara Brook, a Miller Postdoctoral Fellow at UC Berkeley, shared her research seeking to answer this question. Cara started with some basic bat biology that helps explain why they may be so special. Bats are among the longest-lived mammals for their size, are highly gregarious, do that whole flying thing, and have some curious immune functions. Among these are partial resistance mechanisms to viruses that help prevent pathology such as inflammation that is normally associated with viral infection. Even more unique is their constitutive expression of IFN-, an anti-viral cytokine. This suggests that bats are better equipped than other mammals to fight off viral infections, and do so with minimal associated pathology! Seasonality in seroprevalence and viral shedding rates among bats suggests that there may be more to these anti-viral immune responses, though. Cara’s been conducting research in Madagascar in an attempt to identify the precise drivers of this seasonality. She’s found that female fruit bats in Madagascar in particular have increased viral titers during gestation and lactation, perhaps implying that female individuals are sacrificing resources that would normally be devoted to immunity to instead raise some bat babies. Some additional modeling work suggests that maternal immunity passed on to those same bat babies that wanes following the first year of life is also playing an important role in observed seroprevalence patterns. This result was dependent on fitting age-structured models to seroprevalence data from bats of different ages. How do you determine how old a bat is you might ask? Cara, the chiropteran tooth fairy, explained that age can be determined from an extracted bat tooth, and that this simple dental procedure does not affect long-term bat survival or fitness. Moving from the dental to the molecular, Cara next told us about her within-host research that seeks to explain how bats manage the dance between viral exposure, a constitutive viral immune response, and immunopathology. She’s using in vitro experiments and within-host, cell-based models to explore how viruses move between cells and between bats. Cara told us that these results suggest that bats’ constitutive immunity causes higher replication rates of viruses within individuals. It was a tour-de-force of field, modeling, and molecular work and taught everyone here at Berkeley EEID a lot about bats as reservoirs for emerging infectious diseases! Writing by Chris Hoover
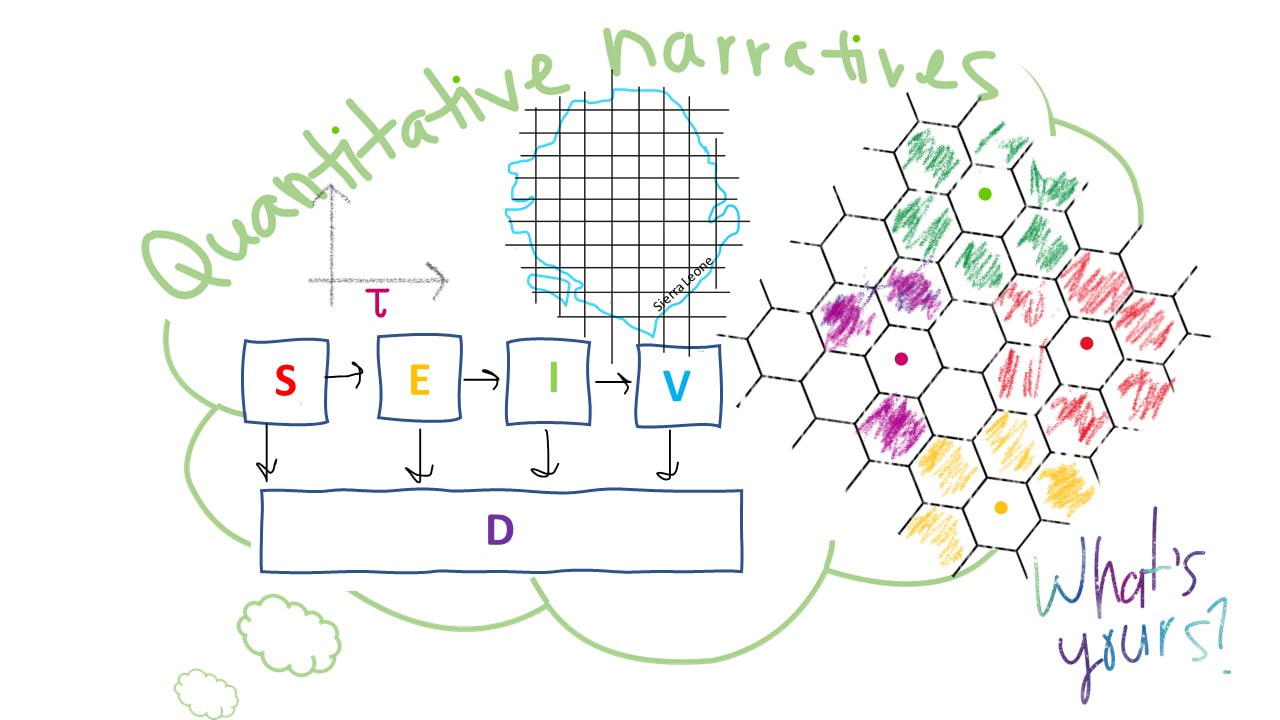
Illustration by Whitney Mgbara Science as narrative in infectious disease biology It’s been a few weeks since we’ve posted a new blog, and that delay is largely due to my tardiness—I was caught up in a grant deadline without a second to spare until now. Apologies! But a bit of time for reflection is rarely a bad thing, and it’s refreshing to revisit this talk now. On Tuesday, September 17, Professor Wayne Getz from UC Berkeley’s Department of Environmental Science, Policy, and Management presented about “Quantitative narratives in disease ecology and the recent outbreak of Ebola in Sierra Leone.” In Wayne’s own words, the talk was heavy on the “quantitative narratives” and less heavy on the Ebola, but it sure gave us plenty to think about. What is a quantitative narrative, you might ask? We don’t often think about narration as quantitative, but it certainly is. Wayne’s talk focused mostly on the fitting of mechanistic models to time series data in ecology and epidemiology, and mechanistic modeling is, by its very nature, narrative. In disease ecology, we distinguish between two broad classes of model: statistical and mechanistic. In statistical models, like the well-known linear regression, data drives the story; we plot two variables against one another and use statistical methods to demonstrate associations between those variables. In mechanistic models, we attempt to describe process, or causation, rather than correlation. This makes the approach much more active, much more narrative. All mechanistic models incorporate a process-based thesis—essentially a narrative—and we test how well each model, each narrative, recaptures the data to which we compare it. As Wayne said himself, “You can never actually prove something is true by fitting a model to data.” But if the model can convincingly recapitulate the data, we tend to accept the narrative on which the model was based. Wayne opened his talk with a broad overview of mechanistic modeling in ecology, touching on many systems that I have not reflected on much since my qualifying exam, including predator-prey models and critical community sizes for population extinction. Then, he dove into the main thread of his talk on zoonoses. Zoonotic pathogens are pathogens that are transmitted from wildlife reservoirs to human hosts, and Wayne’s research group works on several high profile zoonoses: anthrax and Ebola among them. Wayne described another two classes of model—both of the mechanistic type—which his group has used to recapitulate data from zoonotic systems. These model classes—compartmental and individual-based—represent differing approaches to narratives in disease ecology. In compartmental modeling, such as the Susceptible-Infectious-Recovered (SIR) framework, hosts are classed into broad epidemic categories, and modelers use equations to track their movements and define transitions between them. In individual-based modeling, distinct individuals are modeled explicitly, allowing for much greater flexibility in incorporating heterogeneity (i.e. in behavior or immunity) in the system. The decision over what type of model to apply to a problem is a narrative one, as different models tell different stories. My committee member in grad school, Princeton Professor Bryan Grenfell, used to say, “If you apply a complex model to a complex problem, then you have two things that you don’t understand.” Bryan was a big fan of simple compartmental models; simplifying the modeled system down to its fundamental elements tends to give the modeler much greater power to interpret the most important drivers of disease and assess how an effective intervention might be applied. Bryan also worked on a simple disease: measles, a perfectly-immunizing childhood infection that makes it easy to ignore heterogeneities between hosts because most people respond to the virus in very similar ways. In Wayne’s case, he showed us how compartmental models fit to the 2014 Ebola epidemic in West Africa failed to recapitulate the data under the simplest assumptions but did better once he incorporated a time-varying transmission rate. Only by allowing for the transmission rate to slow in the latter half of the epidemic was Wayne able to recapture these data. In his narrative, this is because human behavior changed—contact rates diminished as people stayed home out of fear. It’s a good story, but as Wayne acknowledges himself, you can never really prove it. Wayne closed by introducing us to a new platform in individual-based modeling that his lab group as developed with a software called Nova. Wayne is clearly excited about all of the gadgets embedded in the platform—it allows you to easily incorporate a within-host model (representing immune dynamics, for instance) within a compartmental model and simulate across many layers all via an accessible graphical interface. While certainly offering greater flexibility than a compartmental system, these models are a bit harder to interpret, and fitting these models to data can prove additionally challenging. Ultimately the decision over what type of model to use comes back to the research question and the story that you want to tell. If you are trying to recapitulate data with a credible narrative, then your model will need to produce its own data to compare with reality. Individual-based models hold enormous promise in application to longitudinal cohort studies where the modeler possesses data on individual hosts resampled over time—possibly even including genetic information about pathogen evolution within those hosts. In other cases, where data are less well-resolved, a compartmental approach might be more appropriate. In sum, a mechanistic model is a narrative, the disease ecologist’s way of telling a story. It’s the modeler’s challenge to find the model that is most appropriate for the narrative he or she wishes to tell. Writing by Cara Brook
|
AuthorBerkeley EEID Group Archives
February 2020
Categories
All
|
 RSS Feed
RSS Feed
