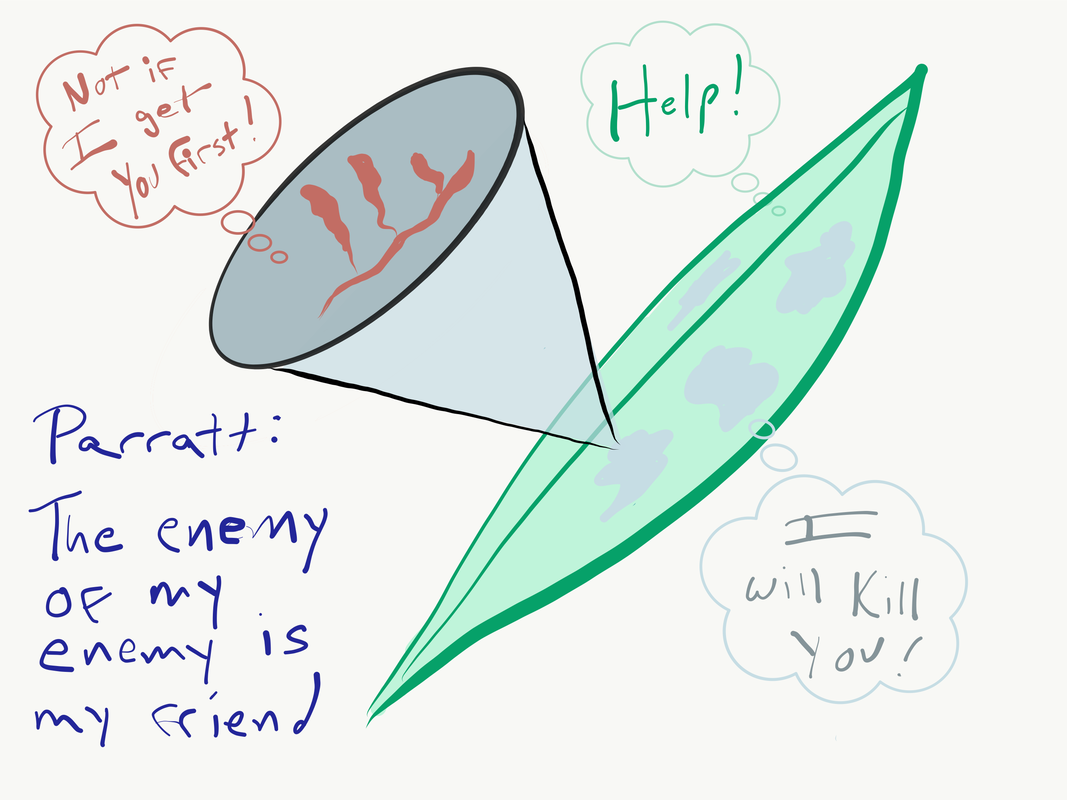
Steve Parratt presents “The enemy of my enemy: the impact of a hyperparasite on a plant pathogen"9/17/2019 Illustration by Britt Koskella This week in the Berkeley Ecology and Evolution of Infectious Disease Seminar, we heard from Dr. Steve Parratt (University of Liverpool) on his work with hyperparasitism. Hyperparasitism, for the unfamiliar, is the phenomenon in which a parasite is itself parasitized by another organism. Hyperparasites can modify the outcome of interactions between host and parasite in unique and surprising ways. Steve started off with a quote from Jonathan Swift illustrating that people have been aware of this concept for some time.
So, naturalists observe, a flea Has smaller fleas that on him prey; And these have smaller still to bite 'em, And so proceed ad infinitum. He then introduced the two systems that he would cover, including work from his PhD and current PostDoc. The first of these projects focused on Arsenophonus nasoniae, a bacterium that parasitized the wasp Nasonia vitripennis which in turn was a parasite of Filth Flies. These bacteria are generally believed to undergo vertical transmission from mother to offspring, and have the interesting effect of killing a large proportion of the male offspring. This makes sense given their assumed mode of vertical transmission, which leaves males as a dead-end host. This also has the intentional or unintentional effect of increasing female fitness through a reduction in competition and decreased chance of inbreeding (as siblings occupy and develop on the same host). However, contrary to the assumption that vertical transmission is the primary route this parasite utilizes, Steve’s work showed that host contact and horizontal transmission were more important in the bacterium’s spread. In cases where horizontal transmission was possible the hyperparasite increased towards fixation and often caused extinction of the local population. This research calls into question the assumption that male-killing is a consequence of vertical transmission, when it may instead be due to benefits conferred during super-infection. When multiple parasite broods infect the same host (super-infection) the parasite has a higher likelihood of transmitting horizontally. In this scenario there is also more likely to be overcrowding, and so the male-killing trait may instead function simply to reduce competition between potential hosts, increasing host and personal fitness. Steve then introduced his current project; investigating the interactions between the fungal plant pathogen commonly known as Powdery Mildew and its hyper-parasite Ampelomyces quisqualis. A. quisualis is believed to negatively impact the probability of Powdery Mildew overwintering, and so Steve set out to understand the role this hyper-parasite might play in the plant-pathogen interaction. First, he showed that the hyper-parasite is variably host specific, with some strains showing evidence of local adaptation and other not. He then found that while the hyper-parasite is common at the meta-population scale it is not especially prevalent in any given population. Within a population A. quisualis seemed to be clustered within a small range of Powdery Mildew infected plants, leading to the conclusion that it is dispersal limited. When investigating the extent to which the hyper-parasite effects Powdery Mildew Steve found that it has small but significant impacts in a lab setting, but no discernible impact in natural experiments. In fact, the only time that A. quisualis had a noticeable impact of Mildew survival was during the Powdery Mildew spore stage. Finally, Steve showed that A. quisualis had a weak but significant effect during co-infection with multiple Powdery Mildew strains, but that this effect did not alter the outcome of competition. Ultimately, this research calls into question the assumption that the “enemy of my enemy is my friend”, as it seems that A. quisualis is either unable, or “un-willing” to exert a meaningful impact on the Host vs. Primary Parasite interaction. In some cases it seems that, as Steve put it: The enemy of my enemy isn’t necessarily a friend if the enemy (and consequently the hyper-parasite) do better when the host does worse. Clearly hyperparasitism is capable of impacting some host parasite interactions, but it may not be safe to assume that’s always the case. This concludes another week of the Berkeley EEID seminar, stay tuned for next week when we hear from Wayne Getz about “Quantitative narratives in disease ecology and the recent outbreak of Ebola in Sierra Leone”. Writting by Eli Mehlferber
12 Comments
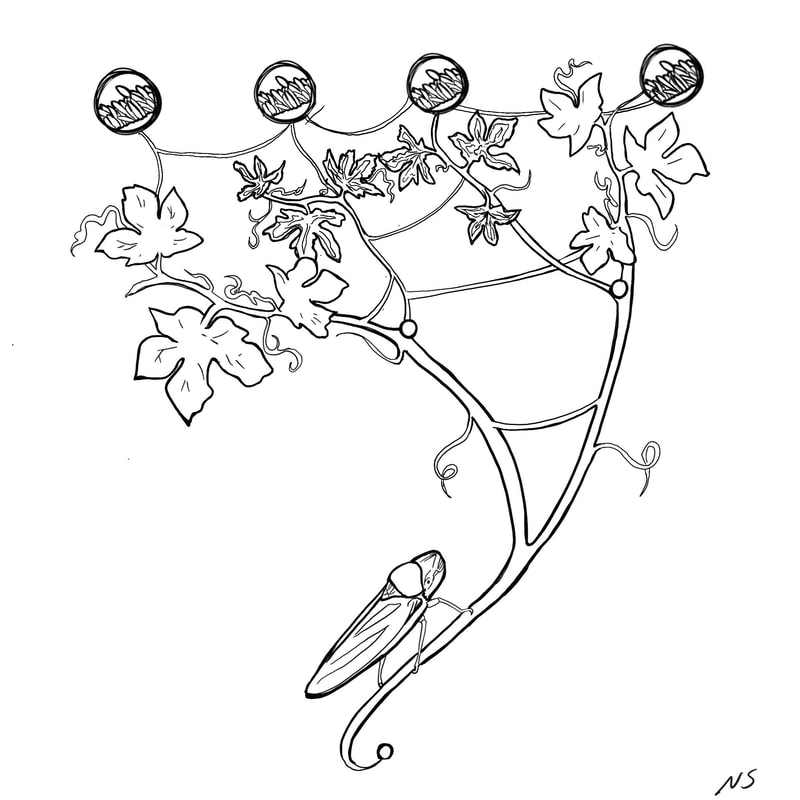
Illustration by Nina Sokolov We kicked off the first official Berkeley Ecology and Evolution of Infectious Disease Seminar with a talk by Professor Rodrigo Almeida from ESPM on his lab’s work on Xylella fastidiosa genetics in agricultural systems. Starting with an introduction to the system, Rodrigo outlined the biology of X. fastidiosa as a multi-host, multi-vector pathogen with high rates of recombination. X. fastidiosa is transmitted by leafhopper vectors that feed on the xylem sap of many plants and can cause severe epidemics and disease in agricultural systems. It has caused large epidemics both historically and recently, so its management is hugely important to people’s livelihoods and local and regional economies. Particularly interesting to my own interests of niche breadth evolution, Rodrigo let us know that this is a highly generalist pathogen, vector, and host complex. In natural ecosystems, he proposes that X. fastidiosa populations are likely composed of many genetically diverse strains that are transmitted by multiple vector species onto multiple host plant species, leading to high levels of strain mixing and recombination. In agricultural systems, there is often one strain of X. fastidiosa being transmitted by one vector species to single host agricultural monocultures. Interestingly, there is no specificity between what bacterial strains colonize which vectors, and the vectors are generalists too, but lots of specificity between bacterial strain and host plant when it comes to disease. Getting into his lab’s recent projects, Rodrigo outlined some of the work that post-doc Andreina Castillo Siri has done to look at how the high recombination rates of X. fastidiosa is associated with disease emergence into new host plants and environments. Rodrigo showed evidence that there is lots of gene flow between introduced and local native X. fastidiosa strains at the start of novel epidemics and shared some initial hypotheses for how recombination early in epidemics may help local adaptation, leading to recombined strains spreading early in epidemics, followed by recombination slowing down as the epidemic ages and there is more ecological isolation. The lab has started to explore this idea by comparing whether X. fastidiosa strains are donors or recipients of genes from both ancestral and recent recombination events in order to identify genes that have undergone recombination and may have been important for recent epidemics. The lab has also worked on historical outbreaks of X. fastidiosa, such as one in California grapevines since the late 1800s. This epidemic spread through California quickly, and, despite few geographic barriers to gene flow, has a highly geographically structured phylogeny with local diversification. These local strains of X. fastidiosa also appear to be recalcitrant to introduction of strains from other Californian regions, suggesting that they may be adapted to their local environments. Rodrigo presented work from Mathieu Vanhove exploring the landscape genomics of X. fastidiosa and how some SNPs correlated with temperature variation. Overall, Rodrigo’s presentation was an exciting start to the seminar series and the diversity of the work he presented highlighted the overlap between the all of our research groups. After a couple of questions, we wandered over to Raleigh’s for Happy Hour to turn our focus towards beers and catching up after the summer! Writing by Elisa Visher
|
AuthorBerkeley EEID Group Archives
February 2020
Categories
All
|
 RSS Feed
RSS Feed
